Quantum Entanglement stabilizes the DNA Molecular Structure
Van der Waals force
In physical chemistry, the van der Waals force (or van der Waals interaction), named after Dutch scientist Johannes Diderik van der Waals, is the sum of the attractive or repulsive forces between molecules (or between parts of the same molecule) other than those due to covalent bonds or to the electrostatic interaction of ions with one another or with neutral molecules. The term includes:
-
force between two permanent dipoles (Keesom force)
-
force between a permanent dipole and a corresponding induced dipole (Debye force)
-
force between two instantaneously induced dipoles (London dispersion force)
It is also sometimes used loosely as a synonym for the totality of intermolecular forces. Van der Waals forces are relatively weak compared to normal chemical bonds, but play a fundamental role in fields as diverse as supramolecular chemistry, structural biology, polymer science, nanotechnology, surface science, and condensed matter physics. Van der Waals forces define the chemical character of many organic compounds. They also define the solubility of organic substances in polar and non-polar media. In low molecular weight alcohols, the properties of the polar hydroxyl group dominate the weak intermolecular forces of van der Waals. In higher molecular weight alcohols, the properties of the nonpolar hydrocarbon chain(s) dominate and define the solubility. Van der Waals-London forces grow with the length of the nonpolar part of the substance.
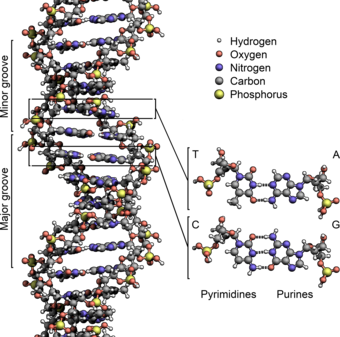
The Relevance Of Continuous Variable Entanglement In DNA
There was a time, not so long ago, when biologists swore black and blue that quantum mechanics could play no role in the hot, wet systems of life.
Since then, the discipline of quantum biology has emerged as one of the most exciting new fields in science. It’s beginning to look as if quantum effects are crucial in a number of biological processes, such as photosynthesis and avian navigation …
(Source: Quantum Entanglement Holds DNA Together, Say Physicists)
The new paradigm is called “Quantum Biology” and the evidence for quantum phenomenons occurring in “living things” at “room temperatures” is overwhelming. There is no way to deny how deep and profound the importance of quantum mechanics are in biological systems.
The structure of the DNA (helix) would vibrate and shake itself apart in a context where just pure classical laws of physics would reign. But all “base pairs” are hold together by “phonons“, which exists in a superposition of states and become entangled, just like other quantum objects. So the result is that the vibration is zero because of the superposition state of all phonons in the DNA helix. They form a kind of “standing wave“.
Please note that quantum mechanics is strictly related to the conscious observer. This has deep implication regarding the maximum life span for humans.
Further reading: